Abstract
Cross-sex hormone treatment is an important component in medical treatment of transsexual people. Endocrinologists are often faced with designing treatment recommendations. Although guidelines from organizations, such as the Harry Benjamin International Gender Dysphoria Association, have been helpful, management remains complex and experience guided. We discuss the range of treatment used by transsexual people, the rationale behind these, and the expectation from such treatment. Recommendations from seven clinical research centers treating transsexual people are discussed. In addition, self-reported hormonal regimens from 25 male-to-female transsexual people and five female-to-male transsexual people are reported. Finally, the potential adverse effects of cross-sex hormone treatment of transsexual people are reviewed. In light of the complexity of managing treatment goals and adverse effects, the active involvement of a medical doctor experienced in cross-sex hormonal therapy is vital to ensure the safety of transsexual people.
Authors - Eva Moore, Amy Wisniewski and Adrian Dobs.
Introduction
Endocrinologists are consulted in the management of transsexual people. Cross-sex hormonal treatment is desired by such patients to successfully live as a member of their identified gender. Endocrine treatment provides some relief from the dichotomy between body habitus and gender identity (1). Ideally, this transition should be quick and complete. However, the medical risks of sex steroids are real; thus, medical providers are confronted with the difficult dilemma of balancing medical risks and psychological needs of patients.
The prevalence of transsexual people has been determined to be as high as 1 in 11,900 males and 1 in 30,400 females (2). Standards of care for the psychological, endocrinological, and surgical management of transsexual people have been proposed by the Harry Benjamin International Gender Dysphoria Association, Inc. (3, 4). Specific management of hormonal regimens and long-term management, however, remain difficult to navigate. As a result, most physicians depend on observational and anecdotal reports to guide endocrine treatment.
This review will discuss the endocrinological treatment of male-to-female (M->F) and female-to-male (F->M) transsexual people. We present the specificities of cross-sex hormone treatment and the reasonable expected effects of those treatments, followed by a brief discussion of the range of treatments used by transsexual people. Our intent is to provide a rationale to guide the endocrinological care of transsexual people.
Subjects and methods
Literature review
A computerized search of the published literature was performed through PubMed, a search engine that matches keywords to medically related articles, abstracts, and bibliographies. Keywords used for the search included transsexual, transsexual people, transgender, transgendered, cross-sex, phytoestrogen, gynecomastia, spironolactone, cyproterone, tamoxifen, and testosterone. Articles in peer reviewed medical journals that suggested the use of sex steroid hormones in transsexual people were retrieved and reviewed for content, and their references were used to identify other literature of interest. Articles that reported clear dosing information for cross-sex hormonal treatment were evaluated. Drug information was obtained from Micromedex, a database of pharmacological agents, and PubMed to obtain generic names when trade names or European brands were reported. A total of 61 articles fit our criteria for review in this manuscript.
Subjects
In addition to reviewing the literature, we present information on transgender individuals seen in the Johns Hopkins Endocrine Clinic. We report on consecutive patients seen in consultation from 1999Б─⌠2000. On presentation, patients were asked to report the hormones they were currently taking. The generic name was recorded for brand names that were reported by subjects. Occasionally, the term used by a subject was too broad to fit a single generic drug, and our best judgment was used to interpret the dose and formulation used.
Results
Feminizing endocrine treatment regimens of M->F transsexual people
Six clinical centers reported dosing information in medical journals for endocrinological treatment of M->F transsexual people (). Treatment regimens included various forms of estrogen, progestins, and/or antiandrogens. No randomized trials were found.
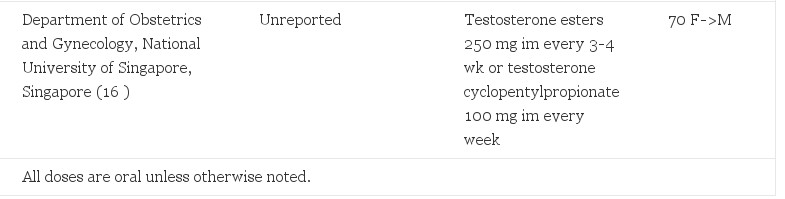
TABLE 1. Hormonal regimens in presurgical transsexual people




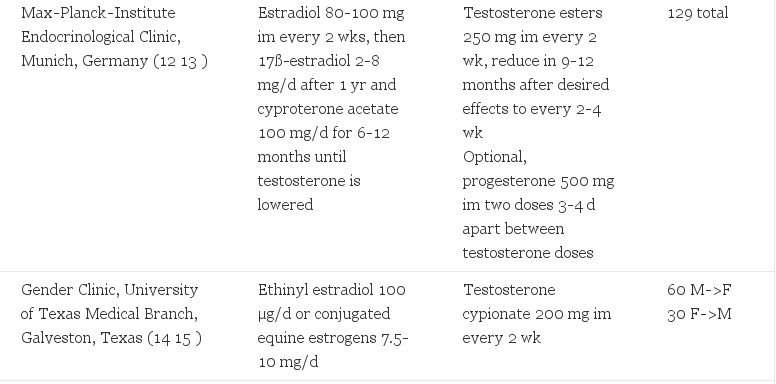
Oestrogens
Estrogen is the cornerstone for feminization of M->F transsexual people. Typical transsexual estrogens were two to three times as high as the recommended doses for hormone replacement therapy (HRT) in postmenopausal women. HRT includes ethinyl estradiol 20Б─⌠50 µg/d, (17) conjugated equine estrogen 0.625 mg/d (18), or transdermal 17ß-estradiol 5 mg/d (19).
Oral delivery was used by most centers. The exception was transdermal 17ß-estradiol in M->F transsexual people over age 40 yr. Transsexual people in The Netherlands were given transdermal estradiol once patients reached 40 yr of age due to an association of thromboembolic events in older transsexual people (6, 20). This policy was supported by a decrease in cardiovascular events after standardizing this regimen in their clinic (6). Of note, im formulations were rarely reported. Avoidance of im dosing was rationalized by the prolonged time to reach steady state and the potential for abuse of this method (21). Additionally, there was no reported sublingual administration of 17ß-estradiol. Pharmacokinetic studies indicate that this route may allow a higher boost of plasma estradiol levels with a greater ratio of estradiol to estrone in comparison to other administrations (22). The safety of this route has not yet been determined.
Higher doses of estrogens and/or im formulations were used in specific conditions for short periods of time. One group applied im 17ß-estradiol 80-100 mg for the initial 6-12 months of treatment (12). Indications include an inability to lower serum testosterone to 50 б╣g/dl (1.74 SI) with no coexisting medical condition or psychiatric clearance for a more rapid feminization (10). Nevertheless, higher doses of estrogens may not lead to more rapid or dramatic clinical changes. Ethinyl estradiol doses of 500 µg demonstrated the same degree of testosterone suppression as 100 µg in a small, observational case-controlled study of M->F transsexual people (14). However, breast growth was enhanced with higher estrogen levels (14). Estrogen doses were lowered in patients with cardiac or other comorbidities (9) or when adverse effects occurred (8, 9, 12, 15, 20). High doses were avoided to minimize adverse effects. After gonadectomy, all centers maintained estrogen therapy. Two centers recommended lowering the dose of estrogens to half the presurgical levels, for example to 50 µg of ethinyl estradiol (6, 9). Alternatively, another center applied a constant hormonal dose both before and after surgery (8). The rationale for continuing estrogen included maintenance of female features and bone mineral density (7, 10).
Antiandrogens and progestins
Concurrent administration of hormonal modulators may potentiate the effects of estrogen. Antiandrogens are theorized to lower serum levels of testosterone or to block its binding to the androgen receptor, thereby decreasing masculine secondary sexual characteristics. Indeed, several studies reported lowering of testosterone with cyproterone acetate 100 µg/d (5, 23). A synergistic effect with estrogen on the physical and emotional changes was also observed by one group with spironolactone (11). This can be particularly helpful in patients with comorbidities that prohibit high levels of estrogen. Cyproterone acetate is not approved for use by the Food and Drug Administration in the United States.
GH-releasing hormone agonists have been considered by some to increase estrogen effects when risk factors limit the dose of estrogen (7).
Addition of progesterone for M->F transsexual people is advocated by some (8, 9, 11). Reasons included enhanced breast growth (9, 11) or to decrease irritability and breast sensitivity (8). However, the clinical effect of progestins was not evident in small observational studies (15). In light of the Womens Health Initiative study (24), progestin therapy should be approached with caution. Combined estrogen and progestin therapy increase the risk of coronary heart disease, strokes, pulmonary embolism, and invasive breast cancers in postmenopausal women on HRT. Possible comparable adverse effects in transsexual people may preclude the empirical use of a progestin for sustained periods of time.
Effects of sexual hormonal treatment in M->F transsexual people
Adverse effects of sex steroid therapy are real and apparent. Retrospective morbidity and mortality data have been reported from the Division of Endocrinology and Andrology at the Free University Hospital in Amsterdam. Of greatest concern is a reported 20-fold increase in venous thrombosis, (6) a decrease from 45-fold in an earlier report (20). Another common phenomenon is an increase in prolactin levels (6, 25) with a possible association with accelerated growth of prolactinomas with feminizing therapy (8, 26, 27). Visual fields and prolactin levels can help assess this risk in patients. Depression is increased in comparison to the general population (20). This is an important reminder that gender reassignment, although effective in relieving the gender dysphoria, should not be considered a cure. Contraindications to therapy have been published and should be considered before initiation of cross-sex hormone therapy (27). Positive and negative effects are reported (Table 2).
Table 2. Side effects of hormonal treatment of transsexual people
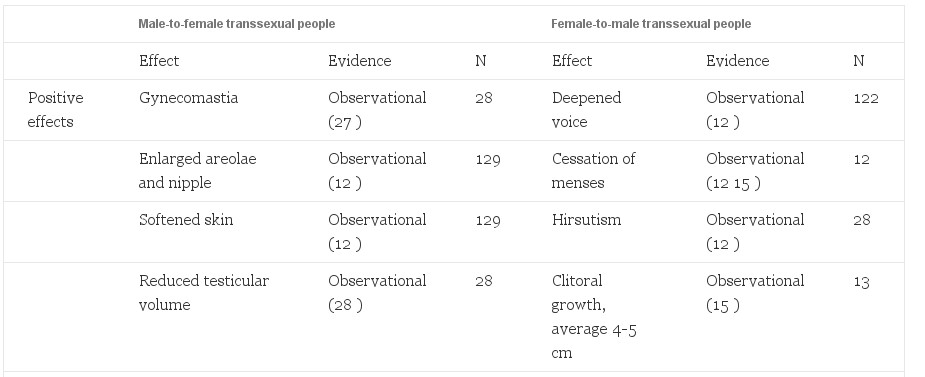
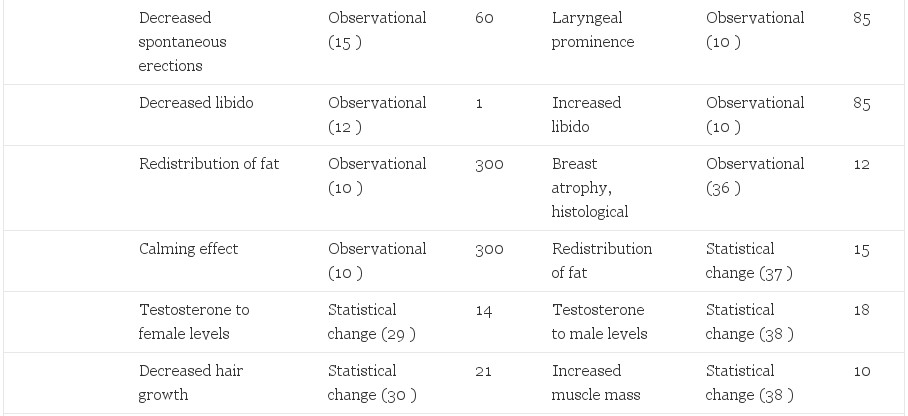
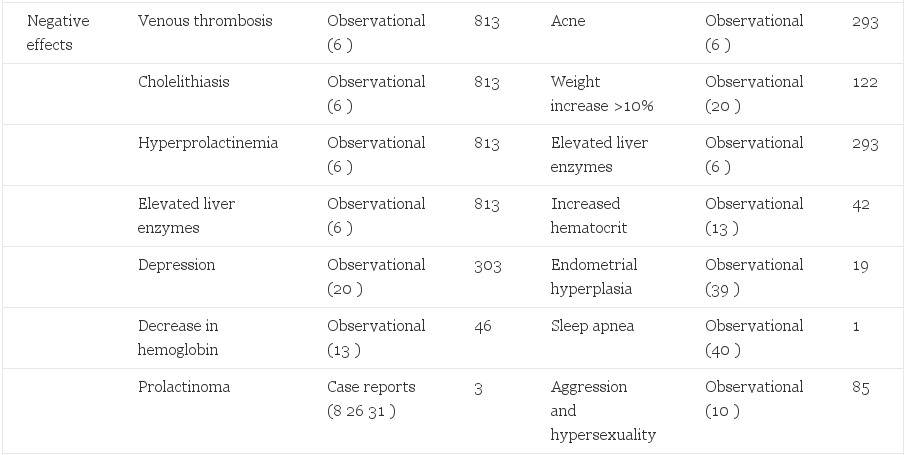
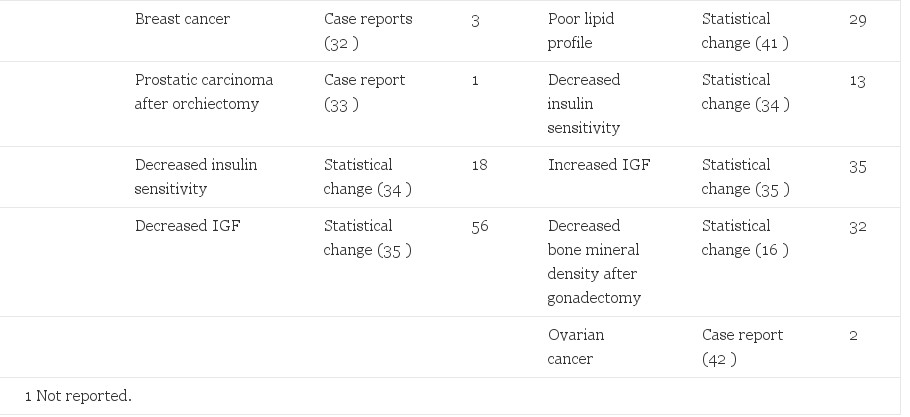
Risks to M->F transsexual people are likely correlated to dose. Estrogen administration to reproductive age women for contraception has demonstrated dose-dependent relationships to venous thromboembolytic disease, pulmonary embolism, myocardial infarction, stroke (43), and adverse liver effects (44, 45). A synergistic risk was seen in women who smoke, are over 35 yr of age, or have other risk factors for cardiovascular disease (46, 47). It is likely that these effects are also present in transsexual people. Smoking cessation, weight reduction, exercise, and appropriate diet are critical elements for preventive health in transsexual people. Minimizing the dose of estrogens, especially in older patients and those with comorbidities, is critical.
Johns Hopkins Clinic self-reported use of feminizing hormones in M->F transsexual people
Description of the Hopkins population
Thirty transgender individuals (97% Caucasian) are reported, including 18 presurgical M->F transsexual people (age, 46.5 yr; range, 39–61 yr), seven postsurgical M->F subjects (age, 49.3 yr; range, 35–68 yr), and five F->M transsexual people (age, 40.2 yr; range, 31–51 yr). The average level of education beyond high school was 3.75 yr. The hormone use presented is that used by the individual upon presentation to the clinic.
Medical regimens used
In contrast to medical recommendations, M->F transsexual people presenting to Johns Hopkins were on much higher doses of sex steroids. Treatment that exceeded recommended estrogen dosages in M->F transsexual people was reported by eight subjects (45%), and five subjects (28%) reported three or more times the recommended dosage. For example, one subject reported taking conjugated equine estrogens 13 mg/d, in addition to a weekly estradiol injection and daily oral medroxyprogesterone. This individual’s dose of conjugated estrogens alone is 21 times the dose recommended for HRT in postmenopausal women. Furthermore, the patients reported using multiple formulations of hormones concurrently. For example, eight subjects (44%) reported taking three or more hormonal medications, and two of those reported five or six different daily medications. Additionally, despite the hesitancy of providers to distribute injectable hormones to M->F transsexual people, 28% reported their use. In light of the older age of subjects, these high doses and complex regimens were particularly concerning for increased risk of adverse effects. It was not asked how the patients obtained these high doses of hormones, but multiple sources were presumed.
Unique to self-reported hormonal regimes, four subjects (22%) reported the use of one or more herbal supplements containing phytoestrogens, although this was not specifically asked. Some studies have demonstrated mild effects of soy isoflavones on male testosterone and estrone levels, although other similar studies have shown no significant change (48). Synergistic possibilities, adverse effects, and drug-herbal interactions are mostly unknown. Phytoestrogens are readily available from the internet and health food stores.
Patients presenting to the endocrine clinic after surgical treatment reported more reasonable hormonal doses. More patients reported hormone dosages similar to those used in hypogonadal women. However, individuals still reported high doses. One reported ethinyl estradiol 0.9 mg/d, oral estradiol 1.25 mg/d, and spironolactone. In comparison, the ethinyl estradiol dose alone is 30 times the dose used in HRT in postmenopausal women. Due to the study design, it is unknown how these estrogen doses compared with previous values in the same individual.
Masculinizing endocrine treatment of F->M transsexual people
Testosterone is the key hormone in F->M transsexual people for the development of secondary sexual characteristics of the desired gender. Injectable testosterone was often used alone, both before and after oophorectomy (Table 1). Oral testosterone undecanoate, available outside of the United States, has been associated with more consistent but lower serum testosterone levels (49). It may not adequately suppress menstruation without the addition of a progestin (12, 20). GH-releasing hormone agonists have been used in adolescent transsexual people to delay puberty, to allow cross-sex hormones to be postponed until adulthood with less psychological stress to the individual (50). Transdermal applications approximated physiological testosterone better than the other delivery methods (51).
Effectiveness was assessed with clinical change, because 17ß-estradiol decreases only minimally (37). Cessation of menses occurred within several months (15). Other effects are reported (Table 2). The appropriate dose of testosterone may vary from patient to patient, but blood levels should be close to the normal mid-male value of 500 µg/dl (17.35 SI). Dosing every 2 wk is recommended to maintain a blood level within physiological range (52).
Adverse effects of masculinizing treatments in F->M transsexual people
Thorough evaluation of the risks of androgen administration to F->M transsexual people has been limited because a small population has presented at treatment centers (6). Retrospective data from 293 patients report no change in mortality, but the population may not be large enough to assess more subtle differences in morbidity and mortality (6).
Adverse effects of androgen administration in F->M transsexual people were observed in two retrospective studies from the Division of Endocrinology and Andrology at the Free University Hospital in Amsterdam. Effects of testosterone administration observed from biological males should be remembered such as polycythemia as a rare complication (53, 54). Contraindications to therapy have been published and should be considered before initiation of cross-sex therapy (55).
Serious adverse risks may be underestimated. The worrisome combination of increased weight, decreased insulin sensitivity, poor lipid profile, and an increase in hematocrit have raised the concern for cardiac and thromboembolytic events. In fact, case reports of cerebral vascular accidents have been reported for individuals with supraphysiological levels of testosterone (56, 57). Polycystic ovarian disease is a risk factor for endometrial cancer (56), and polycystic ovarian morphology of the ovary has been seen in greater numbers in transsexual people before androgen therapy than in the general population (57, 58). Mild endometrial hyperplasia has been appreciated on removal of the uterus (39). A case report detailed two transsexual people with ovarian cancer in F->M transsexual people and raised the question of an association (42). Total hysterectomy after 2 yr of therapy, followed by 50% reduction in testosterone, may be a way to avoid these potential risks, and some experienced clinicians advocated for this procedure (8, 10).
Johns Hopkins Clinic self-reported testosterone use in F->M transsexual people
Five of five F->M transsexual people seen at Johns Hopkins reported testosterone regimens in agreement with published values. Testosterone doses ranged from 150–400 mg/month im. This was similar to the typical dose used in HRT for hypogonadal men [50–400 mg every 2–4 wk (Ref. 59)]. The exact formulations of testosterone used were not clear in most cases, but were likely testosterone enanthate (depot-testosterone) because this is the more readily available testosterone formulation in the United States and the reported doses were consistent with its packaging. Only one subject reported the addition of the antiestrogen, tamoxifen. This subject had a history of breast cancer. The single postsurgical subject reported using a transdermal testosterone patch at 5 mg/d.
Limitations of this review
Limitations to the review of literature include failure to identify articles by the search keywords. Likewise, articles not obviously likely to report hormonal treatment from their titles or abstracts may have been missed. The rationale of hormonal treatments may be more complex than reported in the literature, and tailoring to individual patients may be underreported. The data from surveyed transsexual people might not be an adequate sample of other populations of transsexual people. The results may not be representative of transsexual people of other ages, races, education level, locations, or availability of the internet. Reported medications and doses by transsexual subjects may differ from what is actually taken by subjects. Herbal medications, food choices, and other supplements may have been underreported. The F->M transsexual group in particular may have been too small to adequately represent treatment issues in the general population of F->M transsexual people.
Important comment
Our results show variation among treatment centers with published endocrinological guidelines for the treatment of transsexual people. This is particularly apparent in regard to estrogen dose in people of older ages and the addition of a progestin and/or antiandrogen to the treatment regimen. Results from our survey of M->F transsexual people demonstrated markedly elevated hormone doses and even greater complexity in their treatment regimens. Estrogen doses were often at alarming levels, and multiple formulations were used. This phenomenon has also been observed by other experienced physicians (6, 21, 26, 28, 60, 61). Doses reported by transsexual people were presumed to reflect their personal understanding or experience of what is necessary for their desired physical change. However, to our knowledge no study has evaluated the degree of desired effects seen with these extreme hormonal levels. Modest increases in cross-sex hormones did not show greater efficacy in the few studies available. Instead, other means for more desirable feminine characteristics were advocated by experienced clinicians such as surgery, electrolysis, and/or speech therapy.
Masculinizing therapy for F->M transsexual people was simpler in comparison, with fewer variations between patients and providers. Nevertheless, appropriate dosing was more subjective because clinical markers were better indications of adequate therapy than hormonal levels. Experience has been limited by the small amount of patients that present to treatment centers.
Congruent expectations of transsexual people and providers are crucial to minimize adverse effects. Health risks associated with low-dose sex steroid administration have been reported by the Woman’s Health Initiative (24). Our review suggested that morbidity and mortality associated with cross-sex hormone administration in transsexual people may be associated with much higher risks. Further studies of the long-term implications of cross-sex hormonal administration in transsexual people are needed. Education and close medical monitoring of patients are important aspects of treating transsexual people.
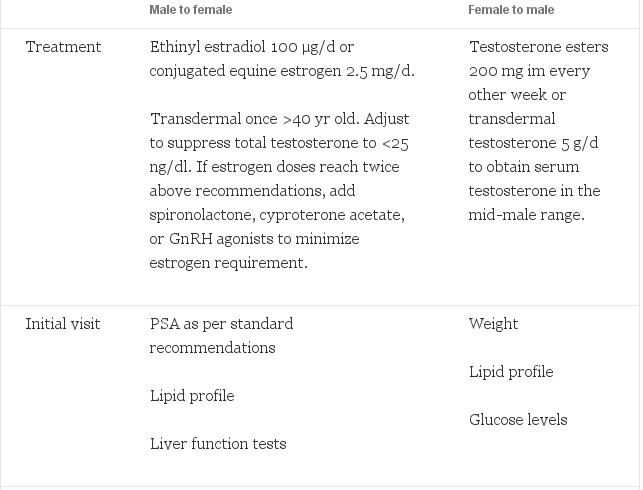
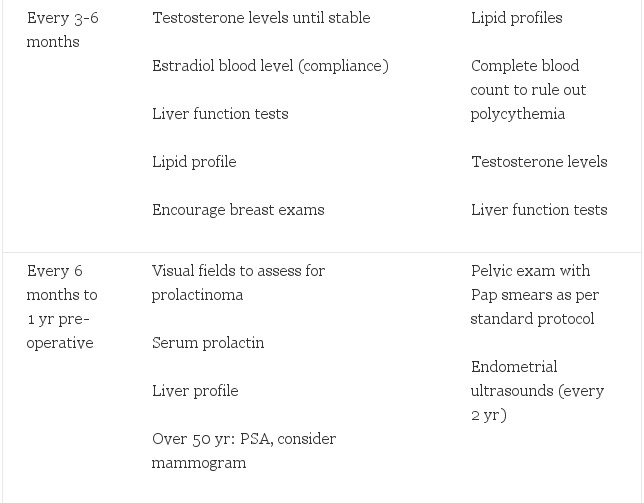
After thorough review of the literature, our recommendations for endocrine treatment and monitoring are reported (Table 3). Feminizing regimens include estrogens used in doses double to those used in hypogonadal women with the goal to suppress testosterone and maintain estrogen at feminine levels. Antiandrogens can be added if testosterone is difficult to suppress with this estrogen dose alone. Some have benefit with the addition of a progestin. Likewise, masculinizing regimens should be similar to that used in hypogonadal men with the goal to keep testosterone in the mid-male range. After gonadectomy, these doses can be substantially decreased while maintaining the above serum levels. Serious adverse effects are apparent in M->F transsexual people. F->M may also experience serious adverse effects, including cardiovascular and gynecological risks. Monitoring of the endometrium by yearly ultrasound is indicated. Any vaginal bleeding after prolonged therapy warrants an endometrial biopsy. Newer, transdermal applications should be considered because they may provide a more consistent hormone level and may decrease severe adverse effects. Close monitoring and yearly reevaluation of treatment are also important to minimize the adverse effects while maximizing the benefits. Age appropriate screening and preventive care for the biological sex of patients is also essential. Randomized clinical trials are needed to build on the years of clinical contributions to this field.
Table 3. Recommended hormonal treatment regimes and follow-up for transsexual people

Acknowledgements
We thank Terry Prendiville, a recipient of a Provost Award from the Johns Hopkins University, for her thoughtful work on this project, as well as Donald E. Moore, M.D., for his help in review of the manuscript.
Footnotes
This work was supported by a Provost Award from Johns Hopkins University.
Abbreviations: F->M, female-to-male; HRT, hormone replacement therapy; M->F, male-to-female.
Received December 13, 2002.
Accepted April 25, 2003.
© Eva Moore, Amy Wisniewski and Adrian Dobs (2003) The Journal of Clinical Endocrinology & Metabolism Vol. 88, No. 8 3467-3473.
References
- Hamburger C 1969 Endocrine treatment of male and female transsexualism. In: Green R, Money J, eds. Transsexualism and sex reassignment. Baltimore: Johns Hopkins Press; 291-307
- Bakker A, van Kesteren PJ, Gooren LJ, Bezemer PD 2002 The prevalence of transsexualism in the Netherlands. Acta Psychiatr Scand 87:237-238
- 2001 The Harry Benjamin International Gender Dysphoria Association standards of care for gender identity disorders, 6th version. http://www.hbigda.org/soc.html. Retrieved November 17, 2002
- Walker P, Berger J, Green R, Laub D, Reynolds C, Wollman L 1985 Standards of care: the hormonal an d surgical sex reassignment of gender dysphoric persons. Arch Sex Behav 14:79-90
- Giltay E, Gooren L, Emeis J, Kooistra T, Stehouwer C 2000 Oral, but not transdermal, administration of estrogens lowers tissue-type plasminogen activator levels in humans without affecting endothelial synthesis. Arterioscler Thromb Vasc Biol 20:1396-1403
- Van Kesteren P, Asscheman H, Megens J, Gooren L 1997 Mortality and morbidity in transsexual subjects treated with cross-sex hormones. Clin Endocrinol (Oxf) 47:337-342
- van Kesteren P, Lips P, Gooren L, Asscheman H, Megens J 1998 Long-term follow-up of bone mineral density and bone metabolism in transsexual people treated with cross-sex hormones. Clin Endocrinol (Oxf) 48:347-354
- Michel A, Mormont C, Legros J 2001 A psycho-endocrinological overview of transsexualism. Eur J Endocrinol 145:365-376
- Futterweit W 1998 Endocrine therapy of transsexualism and potential complications of long-term treatment. Arch Sex Behav 27:209-226
- Leiter E, Futterweit W, Brown G 1993 Gender reassignment: psychiatric, endocrinologic, and surgical management. In: Webster G, Kirby R, King L, Goldwasser B, eds. Reconstructive urology. Boston: Blackwell Scientific Publications; 23:921-932
- Prior J, Vigna Y, Watson D 1989 Spironolactone with physiological female steroids for presurgical therapy of male-to-female transsexualism. Arch Sex Behav 18:49-57
- Schlatterer K, von Werder K, Stalla G 1996 Multistep treatment concept of transsexual patients. Exp Clin Endocrinol Diabetes 104:413-419
- Schlatterer K, Yassouridis A, von Werder K, Poland D, Kemper J, Stalla G 1998 A follow-up study for estimating the effectiveness of a cross-gender hormone substitution therapy on transsexual patients. Arch Sex Behav 27:475-492
- Meyer W, Finkelstein J, Stuart C, Webb A, Smith E, Payer A, Walker P 1981 Physical and hormonal evaluation of transsexual patients during hormonal therapy. Arch Sex Behav 10:347-356
- Meyer W, Webb A, Stuart C, Finkelstein J, Lawrence B, Walker P 1986 Physical and hormonal evaluation of transsexual patients: a longitudinal study. Arch Sex Behav 15:121-138
- Goh H, Ratnam S 1997 Effects of hormone deficiency, androgen therapy and calcium supplementation on bone mineral density in female transsexual people. Maturitas 26:45-52
- Schering Corporation 2002 Estinyl, ethinyl estradiol, USP. In: Walsh P, ed. Physicians desk reference. 56th ed. Montvale, NJ: Medical Economics Company; 3112-3115
- Wyeth-Ayerst Pharmaceuticals 2002 Premarin, conjugated estrogens tablets. In: Walsh P, ed. Physicians desk reference. 56th ed. Montvale, NJ: Medical Economics Company; 3566-3570
- Berlex Laboratories 2002 Climera, estradiol transdermal system. In: Walsh P, ed. Physicians desk reference. 56th ed. Montvale, NJ: Medical Economics Company; 958-962
- Asscheman H, Gooren L, Eklund P 1989 Mortality and morbidity in transsexual patients with cross-gender hormone treatment. Metabolism 38:869-873
- Asscheman H, Gooren L 1992 Hormone treatment in transsexual people. J Psychol Hum Sex 5:39-54
- Price T, Blauer K, Hansen M, Stanczyk F, Lobo R, Bates W 1997 Single-dose pharmacokinetics of sublingual versus oral administration of micronized 17ß-estradiol. Obstet Gynecol 89:340-345
- Jequier A, Bullimore N, Bishop M 1989 Cyproterone acetate and a small dose of oestrogen in the pre-operative management of male transsexual people. A report of three cases. Andrologia 21:456-461
- Writing Group for the Women's Health Initiative Investigators 2002 Risks and benefits of estrogen plus progestin in healthy postmenopausal women. JAMA 288:321-333
- Asscheman H, Gooren L, Assies J, Smits J, De Slegte R 1988 Prolactin levels and pituitary enlargement in hormone-treated male-to-female transsexual people. Clin Endocrinol (Oxf) 28:583-588
- Gooren L, Assies J, Asscheman H, de Slegte R, van Kessel H 1988 Estrogen-induced prolactinoma in a man. J Clin Endocrinol Metab 66:444-446[Abstract]
- Kanhai R, Hage J, Asscheman H, Mulder J 1999 Augmentation mammaplasty in male-to-female transsexual people. Plast Reconstr Surg 104:542-549
- Reutrakul S, Ongphiphadhanakul B, Piaseu N, Krittiyawong S, Chanprasertyothin S, Bunnag P, Rajatanavin R 1998 The effects of oestrogen exposure on bone mass in male to female transsexual people. Clin Endocrinol (Oxf) 49:811-814
- Giltay E, Lambert J, Gooren L, Elbers J, Steyn M, Stehouwer C 1999 Sex steroids, insulin, and arterial stiffness in women and men. Hypertension 34:590-597
- Giltay E, Gooren L 2000 Effects of sex steroid deprivation/administration on hair growth and skin sebum production in transsexual males and females. J Clin Endocrinol Metab 85:2913-2921
- Futterweit W 1980 Endocrine management of transsexual: hormonal profiles of serum prolactin, testosterone, and estradiol. N Y State J Med 80:1260-1264
- Pritchard T, Pankowsky D, Crowe J, Abdul-Karim F 1988 Breast cancer in a male-to-female transsexual: a case report. JAMA 259:2278-2280
- van Haarst E, Newling D, Gooren L, Asscheman H, Prenger D 1998 Metastatic prostatic carcinoma in a male-to-female transsexual. Br J Urol 81:776
- Polderman K, Gorren L, Asscheman H, Bakker A, Heine R 1994 Induction of insulin resistance by androgens and estrogens. J Clin Endocrinol Metab 79:265-271
- van Kesteren P, Lips P, Deville W, Popp-Snijders C, Asscheman H, Megens J, Gooren L 1996 The effects of one-year cross-sex hormone treatment on bone metabolism and serum insulin-like growth factor-1 in transsexual people. J Clin Endocrinol Metab 81:2227-2232
- Futterweit W, Schwartz I 1988 Histopathology of the breasts of 12 women receiving long-term exogenous androgen therapy. Mt Sinai J Med 55:309-312
- Elbers J, Asscheman H, Seidell J, Frolich M, Meinders E, Gooren L 1997 Reversal of the sex difference in serum leptin levels upon cross-sex hormone administration in transsexual people. J Clin Endocrinol Metab 82:3267-3270
- Elbers J, Asscheman H, Seidell J, Megens J, Gooren L 1997 Long-term testosterone administration increases visceral fat in female to male transsexual people. J Clin Endocrinol Metab 82:2044-2047
- Futterweit W, Deligdisch L 1986 Histopathological effects of exogenously administered testosterone in 19 female to male transsexual people. J Clin Endocrinol Metab 62:16-21
- Bardin CW, Swerdloff RS, Santen R 1991 Androgens: risks and benefits. J Clin Endocrinol Metab 73:4-7
- Goh H, Loke D, Ratnam S 1995 The impact of long-term testosterone replacement therapy on lipid and lipoprotein profiles in women. Maturitas 21:65-70
- Hage J, Dekker J, Karim R, Verheijen R, Bloemena E 2000 Ovarian cancer in female-to-male transsexual people: report of two cases. Gynecol Oncology 76:413-415
- Stadel BV 1981 Oral contraceptives and cardiovascular disease. N Engl J Med 305:612–618; 672-677
- Aldinger K, Ben-Menachem Y, Whalen G 1977 Focal nodular hyperplasia of the liver associated with high-dosage estrogens. Arch Intern Med 137:357-359
- Adlercreutz H, Tenhunen R 1970 Some aspects of the interaction between natural and synthetic female sex hormones and the liver. Am J Med 49:630-648
- Hannaford P, Croft P, Kay C 1994 Oral contraception and stroke. Stroke 25:935-942
- Royal College of General Practitioners' Oral Contraception Study 1977 Mortality among oral-contraceptive users. Lancet 2:727-731
- Kurzer M 2002 Hormonal effects of soy in premenopausal women and men. J Nutr 132:570S-573S
- Spinder T, Spijkstra J, Gooren L, Hompes P, van Kessel H 1989 Effects of long-term testosterone administration on gonadotropin secretion in agonadal female to male transsexual people compared with hypogonadal and normal women. J Clin Endocrinol Metab 68:200-207
- Cohen-Kettenis P, van Goozen S 1998 Pubertal delay as an aid in diagnosis and treatment of a transsexual adolescent. Eur Child Adolesc Psychiatry 7:246-248
- Meikle A, Mazer N, Moellmer J, Stringham J, Tolman K, Sanders S, Odell W 1992 Enhanced transdermal delivery of testosterone across nonscrotal skin produces physiological concentrations of testosterone and its metabolites in hypogonadal men. J Clin Endocrinol Metab 74:623-628
- Snyder P, Lawrence D 1980 Treatment of male hypogonadism with testosterone enanthate. J Clin Endocrinol Metab 51:1335-1339
- Wilson J, Griffin J 1980 The use and misuse of androgens. Metabolism 29:1278-1295
- Ammus S 1989 The role of androgens in the treatment of hematologic disorders. Adv Intern Med 34:191-208
- Nagelberg S, Laue L, Loriaux L, Liu L, Sherins R 1986 Cerebrovascular accident associated with testosterone therapy in a 21-year-old hypogonadal man. N Engl J Med 314:649-650
- Futterweit W 1984 Clinical features of polycystic ovarian disease. In: Polycystic ovarian disease. New York: Springer-Verlag; 83-95
- Futterweit W, Weiss R, Fagerstrom R 1986 Endocrine evaluation of forty female-to-male transsexual people: increased frequency of polycystic ovarian disease in female transsexualism. Arch Sex Behav 15:69-78
- Balen A, Schachter M, Montgomery D, Reid R, Jacobs H 1993 Polycystic ovaries are a common finding in untreated female to male transsexual people. Clin Endocrinol (Oxf) 38:325-329
- BTG Pharmaceuticals 2002 Delatestryl, testosterone enanthate injection, USP. In: Walsh P, ed. Physicians desk reference. 56th ed. Montvale, NJ: Medical Economics Company; 1151-1154
- Cooper M 1983 Hormone treatment clinic for transsexual people. Hawaii Med J 43:142-146
- Schaefer L, Wheeler C, Futterweit W 1995 Gender identity disorders (transsexualism). In: Gabbard G, ed. Treatments of psychiatric disorders. 2nd ed. Washington, DC: Am Psych Press; 2015-2079
Comments
comments powered by Disqus